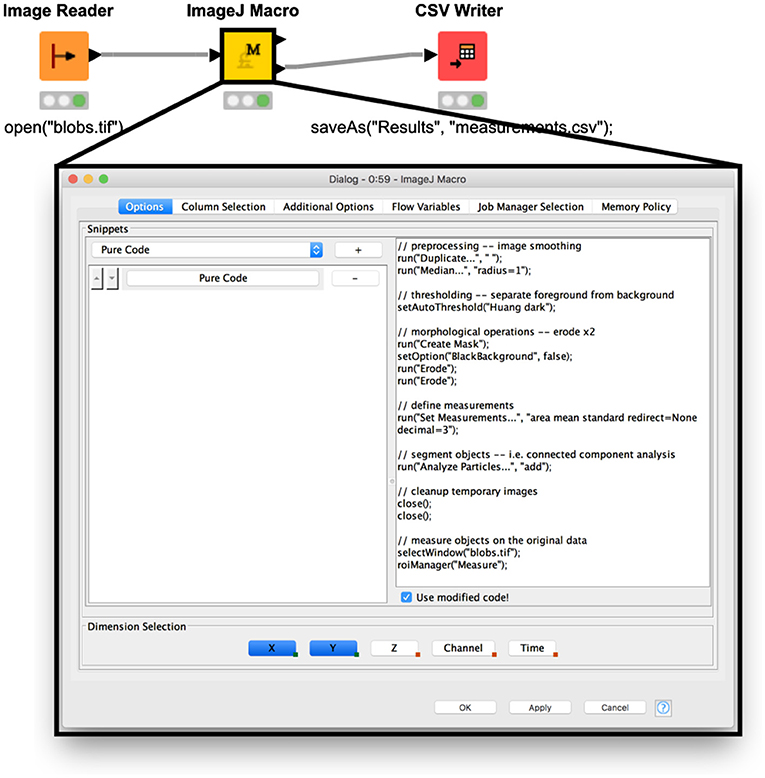
#Nih imagej software function how to
Finally, you will get to know some different ImageJ plugins and will learn how to implement your own. You will also find the instructions necessary to record all the steps you perform so they can be saved and re-run on the same image to ensure analysis reproducibility. You will also learn how to make modifications through ImageJ filters and how to make local measurements using the selections system. Image Processing with ImageJ will start by showing you how to open a number of different images, become familiar with the different options, and perform simple analysis operations using the provided image samples. ImageJ is an excellent public domain imaging analysis platform that can be very easily used for almost all your image processing needs. Image Processing with ImageJ is a practical book that will guide you from the most basic analysis techniques to the fine details of implementing new functionalities through the ImageJ plugin system, all of it through the use of examples and practical cases. ImageJ’s long history and ever-growing user base makes it a perfect candidate for solving daily tasks involving all kinds of image analysis processes. The method and program are validated using the analysis of the spatio-temporal interactions between a G-protein coupled receptor, the tachykinin NK2 receptor, and the beta-arrestin 2 as an example.Digital image processing is an increasingly important field across a vast array of scientific disciplines. It is particularly adapted when transient expression of the fluorescent proteins is used thereby giving very variable expression levels or when the colocalization of the two partners is varying in proportion, in amount, and in size, as a function of time. This method called "FRET and Colocalization Analyzer" has been implemented in a Plug-in of the freely available ImageJ software. Finally, it proposes an alternative to normalization of the FRET intensities to compare FRET signal variations between samples. It displays FRET images as a function of the colocalization of the two fluorescent partners. It proposes imaging treatments and the display of control images to validate the BT calculation and the image corrections.
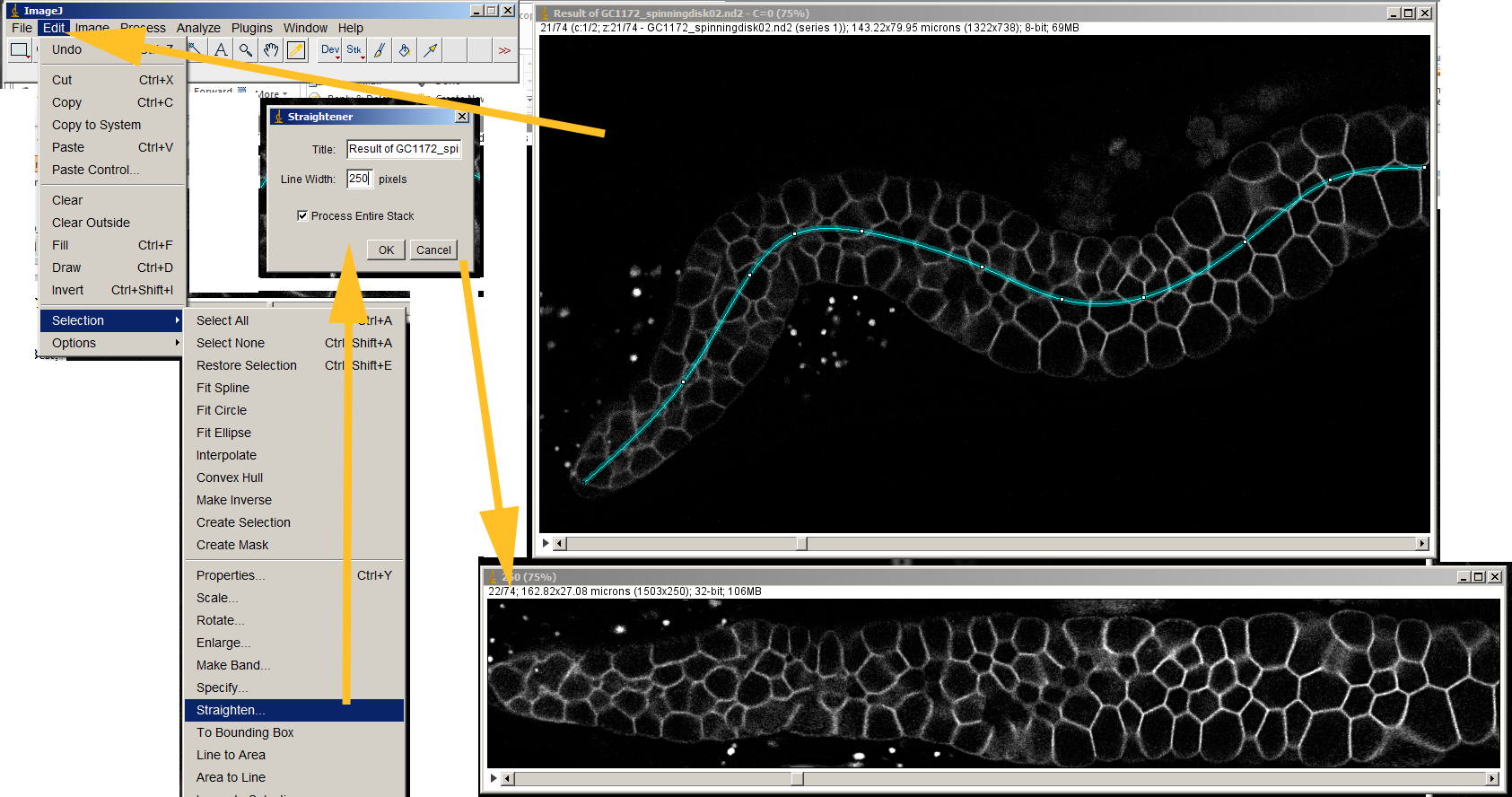
Our method reduces the interference of the user to a minimum by analyzing the entire image, pixel by pixel. Authentic FRET signal measurements require the correction from the FRET channel of the undesired bleed-through signals (BT) resulting from both the leak-through of the donor emission and the direct acceptor emission. We present a method for visualization of FRET images acquired by confocal sensitized emission, involving excitation of the donor fluorophore and detection of the energy transfer as an emission from the acceptor fluorophore into the FRET channel. Fluorescence resonance energy transfer (FRET) between an adequate pair of fluorophores is an indication of closer proximity than colocalization and is used by biologists to study fluorescently modified protein interactions inside cells.
